Under CEPI 3.0, our work is organised around three priority areas: strengthening vaccine development across the pathogens and viral families most likely to spark new outbreaks; advancing rapid‑response platform technologies that can be adapted quickly to the unknown; and building interconnected global networks for research, manufacturing and regulatory readiness so the world can act as one system when new threats emerge.
A key shift in this new strategy is the move from a pathogen‑by‑pathogen approach to a viral family framework — a more flexible and future‑proof way of preparing for both known threats and Disease X.
We spoke with Dr Mandeep Singh Dhingra, MD, CEPI’s Director of R&D Programmes, to unpack what this change means and its role in CEPI 3.0 and the 100 Days Mission.
How does the vaccines and viral families priority area ensure the next outbreak doesn’t catch the world off guard?
When we talk about preparedness, we’re preparing for two things at once: the known threats we already track and the unknown, a Disease X. The viral family framework helps us do both.
Instead of preparing for one pathogen at a time, we prepare for whole families of viruses. That means building knowledge and countermeasures that could potentially apply across a family, so we’re better positioned if a new virus emerges.
Because viral families are diverse, we focus on areas where our work can have the biggest impact across multiple pathogens and invest in pathways that we believe will most efficiently lead to real-world countermeasures. That's how CEPI 3.0 reduces the risk of being caught off guard.
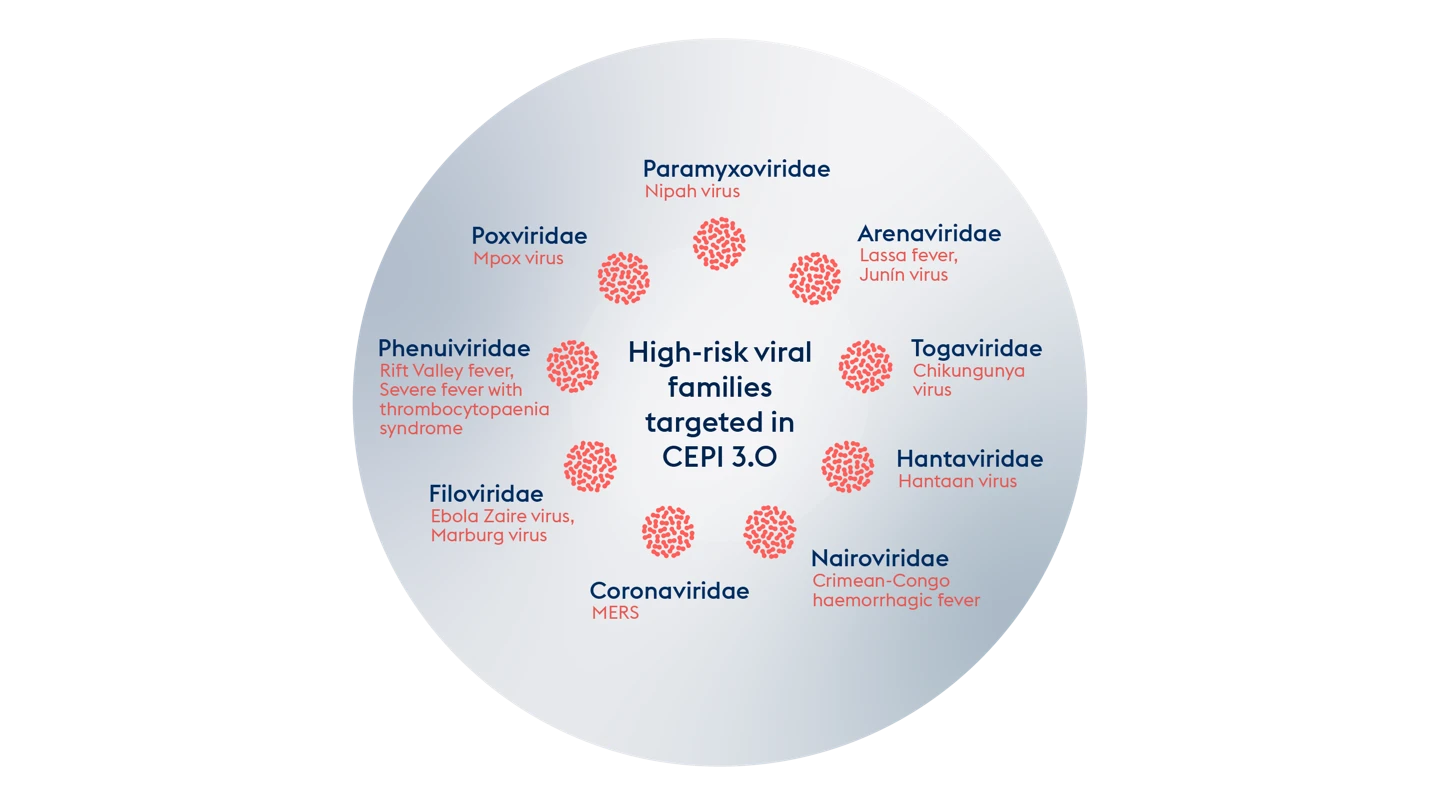
High-risk viral families and example pathogens within the family.
How does this priority area move us from reacting to outbreaks to proactively preparing for them?
First, we’re widening our field of view. Instead of investing in a single pathogen, we ask whether today’s work builds broader readiness and whether insights from one pathogen strengthen our understanding of others, even potentially those from different viral families.
Second, this approach strengthens CEPI’s role as a connector. Preparedness isn’t something CEPI can deliver alone. But we can act as a glue, bringing together the right partners, institutions, and countries. If we can introduce the flexibility and agility that the viral family framework is designed to deliver, it becomes a major step toward proactive global readiness.
At the heart of the CEPI 3.0 strategy is the 100 Days Mission. How does this priority area help to achieve that goal?
The 100 Days Mission is about responding as quickly as possible, where the response is needed most, with tools that make a real difference. The viral family framework helps us get there by shifting our focus from the actual activities that we support to what impact they deliver. I think of it as shifting our philosophy from ‘the what’ to the ‘so what’ and shifting our approach from reaction to readiness. That's why every investment is assessed for its potential to shorten timelines, generate actionable scientific intelligence and support rapid vaccine development.
But the engine isn’t only scientific. Countries and regions must also own and prioritise the 100 Days Mission. CEPI can act as the integrating factor, but success depends on partnership and local execution.
How do we ensure this approach is regionally anchored and not just for high‑income countries?
Equity is already embedded in CEPI’s work, and the viral family framework makes this even more deliberate. Many pathogens we focus on, like Nipah and Lassa, are regionally concentrated, so strengthening research and manufacturing capacity in those regions is essential. It also helps build the self-sufficiency needed to deliver the 100 Days Mission.
Whether that’s working with partners in India to ensure future Nipah vaccine doses can be manufactured regionally, or strengthening partnerships in Africa and Latin America for diseases like Lassa and Chikungunya, respectively, we’re already taking this approach. This means that, say a Disease X emerges from the paramyxoviruses, Nipah’s viral family, in theory, that region would be better prepared to respond at pace.
This regional capability is central to global equity.
How does this work prepare us for a Disease X?
Disease X represents the pathogen we can’t predict, but we can prepare for it both by working directly on knowledge generation and indirectly by using the known to inform the unknown. CEPI is already actively supporting knowledge‑generation work, including the use of artificial intelligence, to help predict which pathogens could emerge and what antigenic structures might generate the strongest immune response.
The viral family framework broadens this readiness. By working across families and tailoring our scientific efforts accordingly, we reduce the likelihood of starting from scratch when the next unknown threat appears.
How does this priority area rely on and strengthen the others?
Everything in CEPI 3.0 is interconnected. The viral family approach depends on and strengthens our work on platforms and networks. You can think of them a bit like a lamp, a lightbulb, and a power socket: each has a role, but only together do they produce light. It’s the combination that creates real capability.
Platforms allow us to quickly adapt vaccine candidates for different viruses within a family in ways regulators can accept. Manufacturing partnerships ensure those vaccines can be produced rapidly and, where possible, regionally. And our networks help move products through development efficiently and anchor everything under the 100 Days Mission.




