Research by CEPI's vaccine R&D team, published in Nature Reviews Drug Discovery, has identified 115 COVID-19 vaccines in development.
The CEPI team have been continuously maintaining an overview of the global landscape of COVID-19 vaccine development activity.
From February, 2020, onwards, the team studied the landscape of COVID-19 vaccine candidates by using internal and external data sources, including vaccine development programmes reported through the WHO's authoritative and continually updated list; information from calls for proposals and direct communications with vaccine developers and partners; clinical trial and funders databases, publicly available literature; and media and press releases.
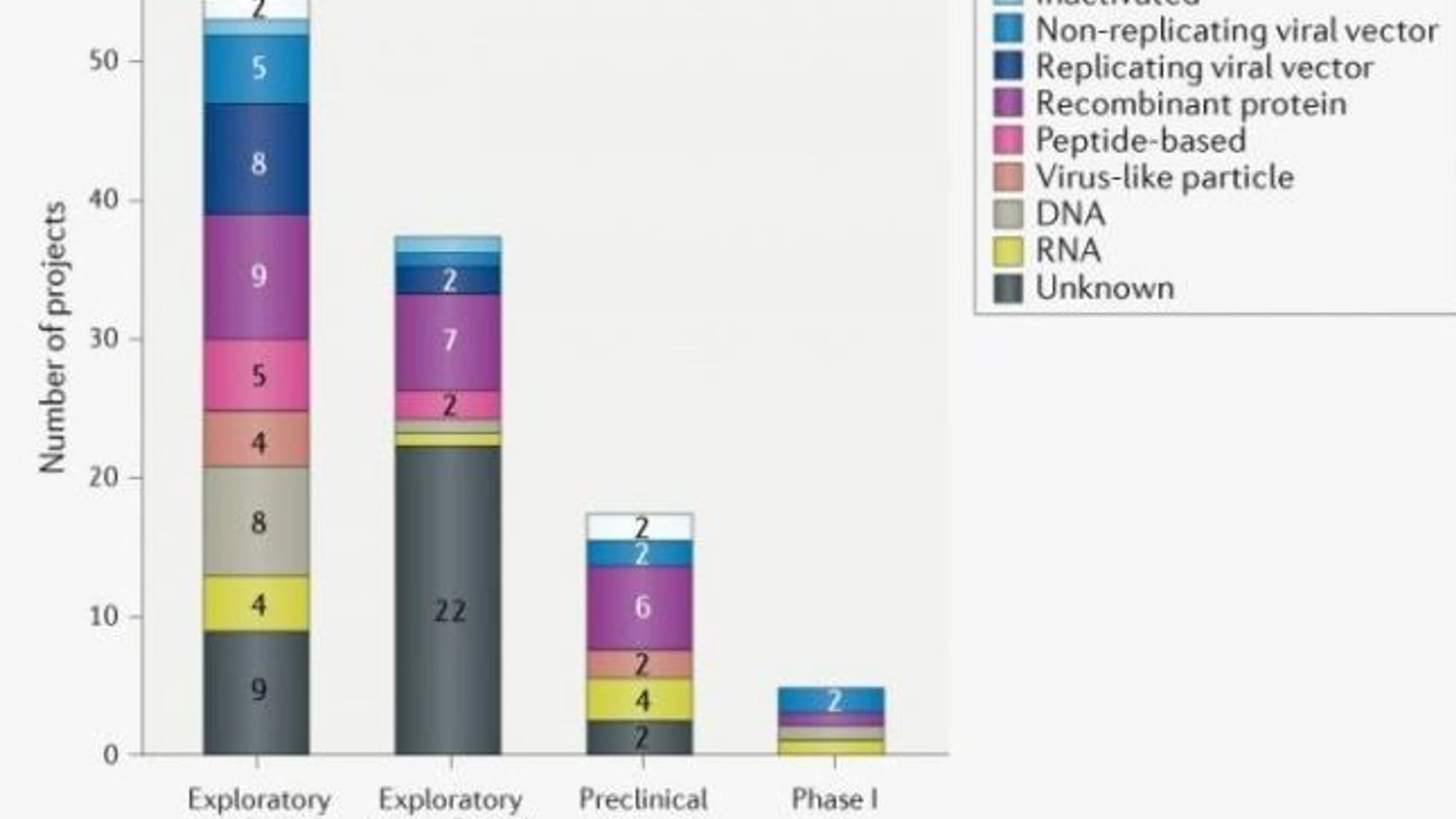
Pipeline of COVID-19 vaccine candidates by technology platform
Wide range of vaccine technology being tested
As of April 8, 2020, 115 vaccine candidates are in varying stages of development. 78 of which are confirmed as active and 37 are unconfirmed (development status cannot be determined from publicly available or proprietary information sources). The study showed that a wide range of technology platforms are being assessed, including traditional and novel approaches.
Most of the active projects are in exploratory or preclinical stages. However, 5 candidates have recently moved into clinical development, including mRNA-1273 (Moderna), Ad5-nCoV (CanSino Biologicals), INO-4800 (Inovio), LV-SMENP-DC and pathogen-specific aAPC (Shenzhen Geno-Immune Medical Institute). Notably, Moderna was able to start clinical testing of its mRNA-based vaccine just 63 days after sequence identification.
The paper also points out that public information on the specific SARS-CoV-2 antigen(s) used in vaccine development is limited and that most vaccine candidates for which information is available aim to induce neutralizing antibodies against the viral surface protein referred to as the "spike protein".
The lead developers
56 (72%) of 78 the confirmed active vaccine candidates are being developed by private or industry developers. 22 (28%) of projects being led by academic, public sector, and other not-for-profit organizations.
Although a number of large multinational vaccine developers (such as Janssen, Sanofi, Pfizer and GlaxoSmithKine) have engaged in COVID-19 vaccine development, many of the lead developers were identified as small and/or inexperienced in large-scale vaccine manufacture.
Geographical distribution of vaccine R&D
Of the confirmed active vaccine candidates, 36 (46%) developers are in North America, 14 (18%) in China, 14 (18%) in Asia (excluding China) and Australia, and 14 (18%) in Europe.
Lead developers of active COVID-19 vaccine candidates are distributed across 19 countries, which collectively account for over three-quarters of the global population.
No public information on vaccine development activity in Africa or Latin America is available. However, it was noted that vaccine manufacturing capacity and regulatory frameworks exist in these regions.
Insights
The authors of this paper stressed that strong international coordination and co-operation among vaccine developers, regulators, policymakers, funders, public health bodies and governments will be needed to ensure safe and effective vaccine can be manufactured in sufficient quantities and made globally accessible.
They also highlighted that CEPI maintains a dynamic approach to the management of its vaccine portfolio and is committed to making its enabling science resources available globally.
Finally, the authors urged the global vaccine community to collectively mobilise the technical and financial support needed to successfully address the COVID-19 pandemic through a global vaccination programme. Earlier this year, CEPI published an estimate that $2 billion would be required to develop a vaccine against COVID-19.
Feature image credit - NIAID
Figure credit - Nature Reviews Drug Discovery