CEPI Awards $25 Million Contract to Profectus BioSciences and Emergent BioSolutions to Develop Nipah Virus Vaccine

OSLO, Norway, BALTIMORE and GAITHERSBURG, Md., May 24, 2018
CEPI—the Coalition for Epidemic Preparedness Innovations—today announced a collaboration with Profectus BioSciences, Inc. and Emergent BioSolutions Inc. (NYSE: EBS) under which Profectus and Emergent will receive up to $25 million to advance the development and manufacture of a vaccine against the Nipah virus, a bat-borne virus that can spread to both humans and livestock.
Under the terms of the Framework Partnering Agreement for the collaboration among the three parties, Profectus will receive development funding from CEPI for advancing its Nipah virus vaccine and Emergent will provide technical and manufacturing support for the CEPI-funded program. Emergent, through a separate agreement with Profectus, has an exclusive option to license and to assume control of development activities for the Nipah virus vaccine from Profectus. The international nonprofit organization PATH will also be working with the consortium under a separate agreement with CEPI to work on clinical development.
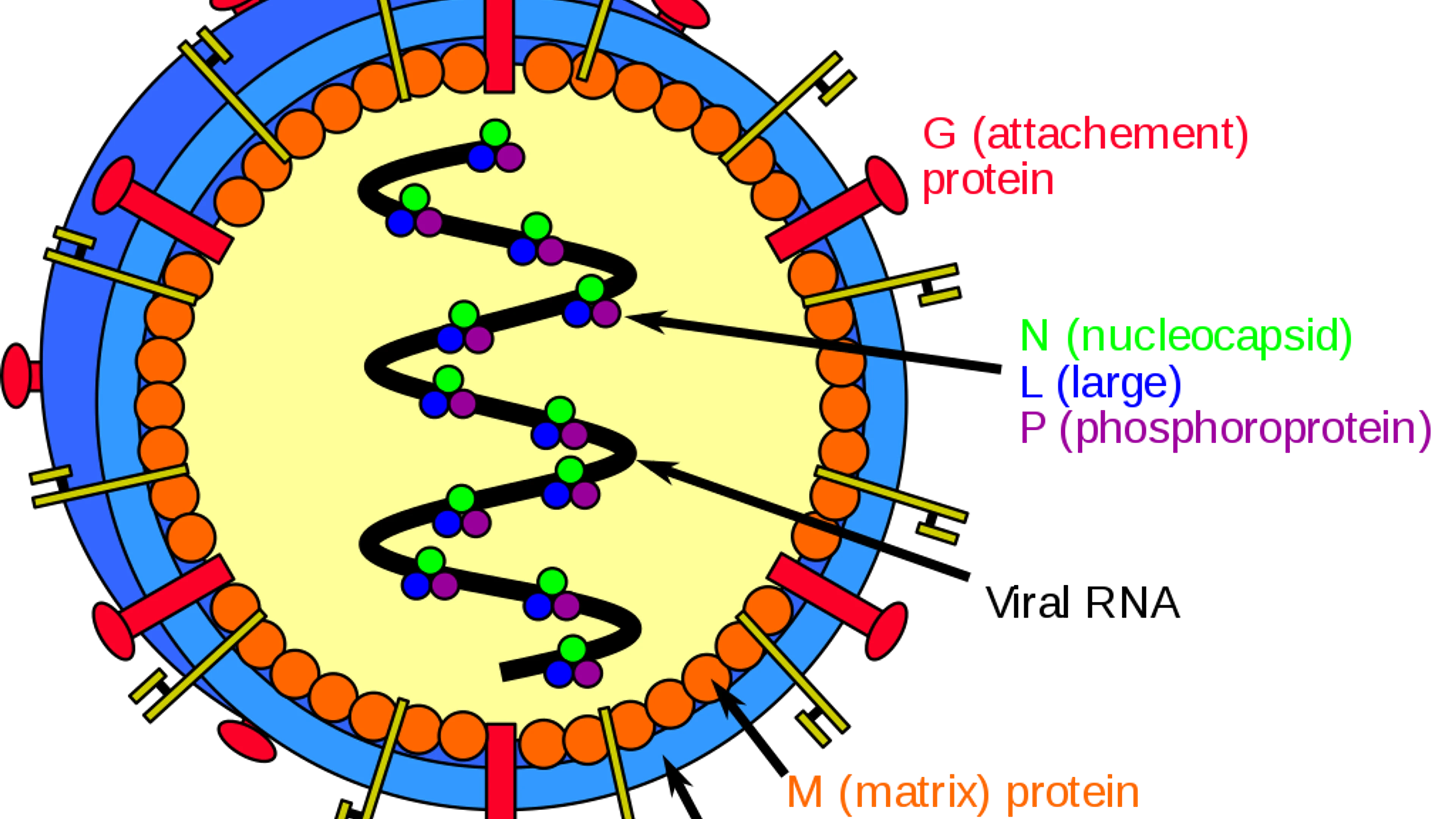
Nipah virus (NiV) and Hendra virus (HeV) are closely related Paramyxoviruses that cause respiratory and encephalitis disease in a variety of animal hosts and in humans. The natural reservoir for these viruses is thought to be flying foxes (bats of the genus Pteropus) found in Southeast Asia and Malaysia. Human infection occurs after direct exposure to tissues and secretions from infected horses, pigs, and bats. There is currently no approved vaccine or therapeutic against either NiV or HeV. The primary treatment for human cases is intensive supportive care, and the mortality rate is high at approximately 75% of cases.
The Southern India state of Kerala is currently in the midst of a Nipah Virus (NiV) outbreak, where the virus has killed 10 people. These are the first documented cases in Kerala.
Previously unidentified, a 1999 NiV outbreak killed 105 people in Malaysia. In addition, more than one million pigs were slaughtered to stem the spread of the virus, which caused significant trade loss and market turmoil. Outbreaks occur annually in Bangladesh and have been reported numerous times in India (CDC). NiV has a 75 percent mortality rate . NiV is shed by bats in Southeast Asia and Malaysia and is transferred to humans via bat urine and saliva on fruit or from infected horses and pigs. It can also spread human-to-human.
The current outbreak of Nipah in India, the government of which is one of CEPI's founders, demonstrates that this is a deadly pathogen that has already travelled thousands of kilometers, has serious epidemic potential and the ability to surprise us. This is the first funding agreement we have signed to develop a vaccine against Nipah and our partnership with Profectus, Emergent, and PATH represents a vital move forward in the global battle against this devastating disease.
The investment of up to $25 million represents an innovative approach to funding vaccine development, unlocking research and development potential so that vaccines are ready for efficacy studies during an outbreak. The agreement will enable funding for development efforts over a five-year period.
Profectus gratefully acknowledges CEPI's leadership and funding of this strategic collaboration to rapidly produce a vaccine against the emerging Nipah virus threat. Nipah's human-to-human transmission has been documented, and any initially small outbreak could take the same course as the recent Ebola outbreak. With CEPI's support and the talents of these highly regarded global health innovation partners, we look forward to applying our expertise and technologies and leveraging our partnership with Emergent to rapidly advance our pre-IND vaccine candidate for Nipah virus as an effective countermeasure that will be positioned to save patients around the world from this devastating disease.
Emergent has a 20-year track record of success working with government and industry partners to provide life-saving medical countermeasures against identified biological agents particularly when a rapid response to an emerging outbreak may be necessary. We look forward to leveraging our expertise in advanced development and manufacturing, including our U.S. government-designated Center for Innovation in Advanced Development and Manufacturing, which is designed to provide surge manufacturing capabilities during public health emergencies. We expect to work closely with Profectus enabling our companies to provide synergistic and complementary capabilities for a successful collaboration with CEPI.
PATH is delighted to partner with CEPI and leverage the end-to-end capabilities of our Center for Vaccine Innovation and Access (CVIA), particularly our success record in clinical development and scaling the introduction of new vaccines in low-resource settings. PATH looks forward to collaborating with CEPI to achieve our shared goals for epidemic preparedness by equipping the world with new tools to protect against these deadly threats.
About the Nipah Vaccine Development
The vaccine in development is based on Nipah virus and Hendra virus technology developed beginning more than 15 years ago by Dr. Christopher Broder and Dr. Katharine Bossart at the Uniformed Services University of the Health Sciences (USU). The vaccine was tested in multiple preclinical models and licensed to Profectus BioSciences by The Henry M. Jackson Foundation for the Advancement of Military Medicine (HJF) to develop the human vaccine. Dr. Broder began a collaboration with Dr. Antony Dimitrov of Profectus BioSciences to develop the human vaccine with financial support from the United States National Institutes of Health. As a next step in the Nipah vaccine development, Dr. Thomas Geisbert, a Professor in the Department of Microbiology and Immunology at The University of Texas Medical Branch (UTMB) at Galveston National Laboratory (GNL), will conduct correlate of immunity studies at its biosafety level 4 facility; and HJF, USU, and Profectus will collaborate on the development of a clinical assay for the evaluation of the anti-Nipah virus vaccine response. PATH is responsible for the clinical development of the Nipah vaccine candidate.
Based on the success of this vaccine technology against Nipah and Nipah's close relative, Hendra virus, it is highly likely that the Nipah vaccine to be developed here will also work against Hendra virus,
About CEPI
CEPI is an innovative partnership between public, private, philanthropic and civil organizations founded in Davos in 2017 to develop vaccines to stop future epidemics. To date, CEPI has received multi-year funding from Norway, Germany, Japan, the Bill & Melinda Gates Foundation, and Wellcome. CEPI has also received single-year investments from the governments of Australia, Belgium, and Canada. It has reached $630 million of its target $1 billion funding target. The European Commission has announced a contribution in kind of €250 million that will support relevant projects through EC mechanisms. Since its launch in January 2017, CEPI has announced two Calls for Proposals. The first was for candidate vaccines against MERS-COV, Nipah and Lassa viruses. The second was for the development of platforms that can be used for rapid vaccine development against unknown pathogens.
About Profectus BioSciences, Inc.
Profectus BioSciences, Inc. is a clinical-stage vaccine platform company developing novel vaccines for the prevention and treatment of infectious diseases and associated cancers. Profectus' vaccines are based on the company's proprietary vaccine delivery platforms to provide protection against emerging infectious diseases of public health and biodefense importance such as Ebola, Marburg, Chikungunya, Zika, the equine encephalitis viruses, and respiratory syncytial virus; and therapeutically targeting virally infected cells and cancers associated with hepatitis B virus (HBV), human papilloma virus (HPV), herpes simplex virus type 2 (HSV-2), and human immunodeficiency virus (HIV). For more information, please visit www.profectusbiosciences.com.
About Emergent BioSolutions Inc.
Emergent BioSolutions Inc. is a global life sciences company seeking to protect and enhance life by focusing on providing specialty products for civilian and military populations that address accidental, intentional, and naturally occurring public health threats. Through our work, we envision protecting and enhancing 50 million lives with our products by 2025. Additional information about the company may be found at www.emergentbiosolutions.com. Follow us on Twitter @emergentbiosolu and Instagram @life_at_emergent.
About PATH
PATH is the leader in global health innovation. An international nonprofit organization, we save lives and improve health, especially among women and children. We accelerate innovation across five platforms—vaccines, drugs, diagnostics, devices, and system and service innovations—that harness our entrepreneurial insight, scientific and public health expertise, and passion for health equity. By mobilizing partners around the world, we take innovation to scale, working alongside countries primarily in Africa and Asia to tackle their greatest health needs. Together, we deliver measurable results that disrupt the cycle of poor health. Learn more at www.path.org.
About the Uniformed Services University of the Health Sciences (USU)
The Uniformed Services University of the Health Sciences (USU), founded by an act of Congress in 1972, is the academic heart of the Military Health System. USU students are primarily active-duty uniformed officers in the Army, Navy, Air Force and Public Health Service who receive specialized education in tropical and infectious diseases, TBI and PTSD, disaster response and humanitarian assistance, global health, acute trauma care, and advanced practice nursing and dentistry. For more information, visit www.usuhs.edu.
About HJF
The Henry M. Jackson Foundation for the Advancement of Military Medicine, Inc. (HJF) is a private, not-for-profit organization authorized by Congress to support military medicine at the Uniformed Services University of the Health Sciences and throughout the armed forces. HJF serves military, medical, academic and government clients by administering, managing and supporting preeminent scientific programs that benefit members of the armed forces and civilians alike. Since its founding in 1983, HJF has served as a vital link between the military medical community and its federal and private partners. HJF's support and administrative capabilities allow military medical researchers and clinicians to maintain their scientific focus and to accomplish their research goals effectively and efficiently. For more information, visit www.hjf.org.
About the Galveston National Laboratory
The preclinical studies will be conducted in UTMB's Galveston National Laboratory, a state-of-the-art high containment biosafety Level 4 facility where scientists wear protective suits and study pathogens that cause severe diseases or are fatal. The GNL provides much needed research space and specialized research capabilities to develop therapies, vaccines and diagnostic tests for naturally occurring emerging diseases such as SARS, West Nile encephalitis, Ebola and avian influenza—as well as for microbes that might be employed by terrorists. UTMB has the only functioning Level 4 laboratory located on an American university campus.
Contacts
CEPI
Rachel Grant
+44(0)7891249190
Profectus BioSciences, Inc.
Mary Moynihan
M2Friend Biocommunications
802-951-9600
Emergent BioSolutions Inc.
Investors:
Robert G. Burrows
Vice President, Investor Relations
240-631-3280
Media:
Lynn Kieffer
Vice President, Corporate Communications
240-631-3391