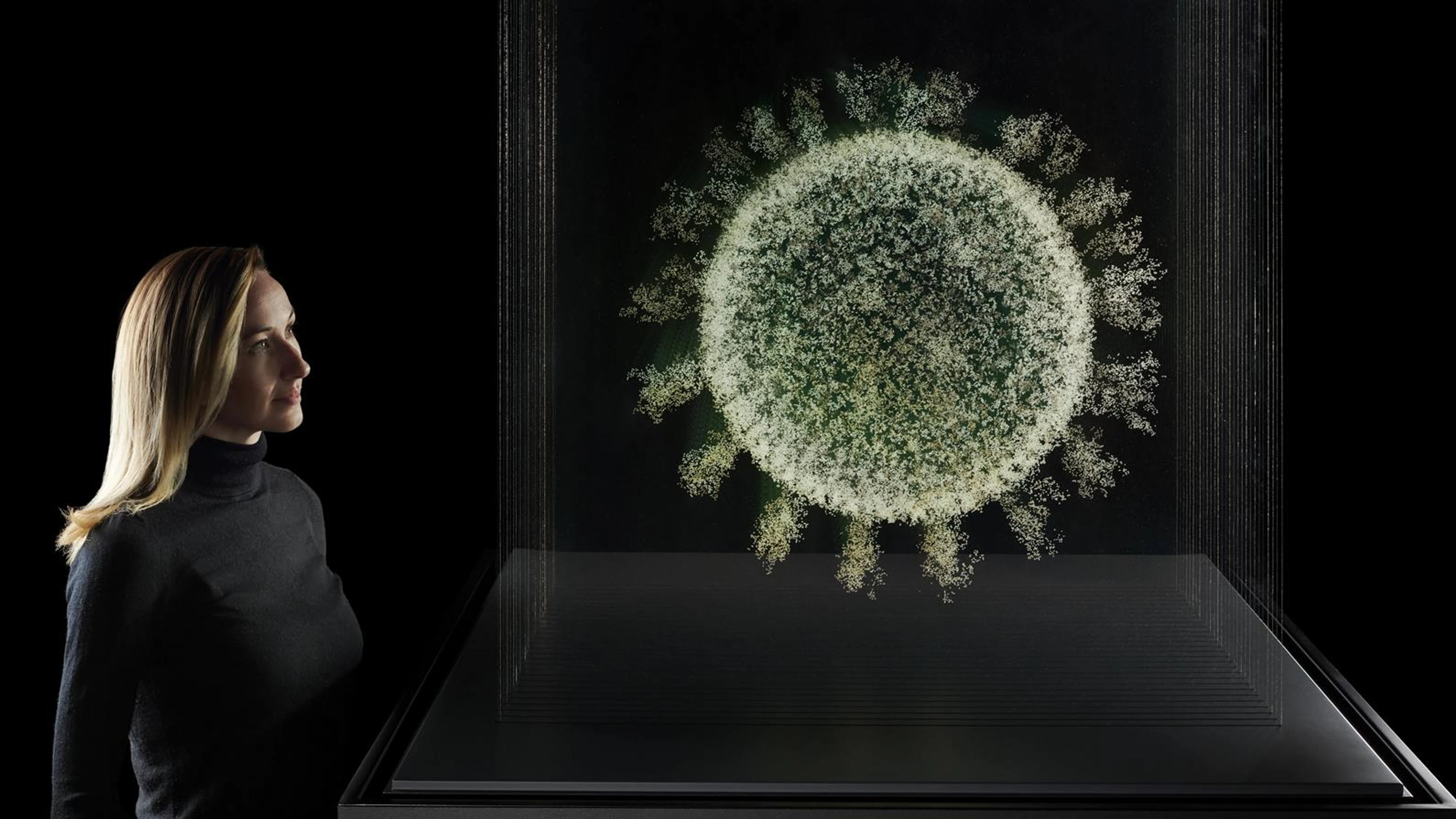
It took just 326 days from release of the genetic sequence of the SARS-CoV-2 virus to the world achieving authorisation of the first COVID-19 vaccine. The Science Museum's latest exhibition, Injecting Hope: The Race for a Covid-19 Vaccine, skillfully describes this remarkable story.
At an event to mark the launch of the exhibition at the Science Museum, Dr Richard Hatchett (CEO of CEPI) sat down with Roger Highfield OBE (Director of Science at the Science Museum) to discuss how CEPI plans to help the world compress vaccine development timelines down even further to just 100 days, regardless of the kind of virus that might emerge in the future.
The interview has been edited for brevity and clarity. Watch the full interview here.
Roger Highfield: Let's put the whole 100 days effort in context. The race for the COVID-19 vaccine seems a really good place to start. Give us your take on how well the global scientific community did in response to outbreak in 2020?
Richard Hatchett: I think the scientific community accomplished amazing things. We developed a vaccine in about 11 months from the release of the genetic sequences, before it was available under emergency use authorization. And that was the fastest that a vaccine had ever been developed, the previous record for developing a vaccine from the isolation of a virus was about four and a half years. And it was a mumps vaccine that was developed in the 1960s. No vaccine had ever been developed faster than that.
What the world accomplished in response to COVID-19 was amazing, it was unprecedented. And it was also insufficient. By the time the vaccines became available, there were tens of millions of cases, the virus had spread all around the world. So we have to develop vaccines much more rapidly in response to future threats, we have to be much more prepared, in many ways beyond just vaccine development for future pandemic threats.
I understand your point that we could have done so much better, but we've still got to keep on pinching ourselves that we managed to deliver several vaccines in about a year…
Not only that, within a year of the first vaccine becoming licensed the world had produced and delivered somewhere on the order of 12 billion doses of vaccine. To put that into perspective, in 2019, the total global production of all vaccine manufacturers of all vaccines was about 5.5 billion doses. So it speaks to how rapidly we scaled up.
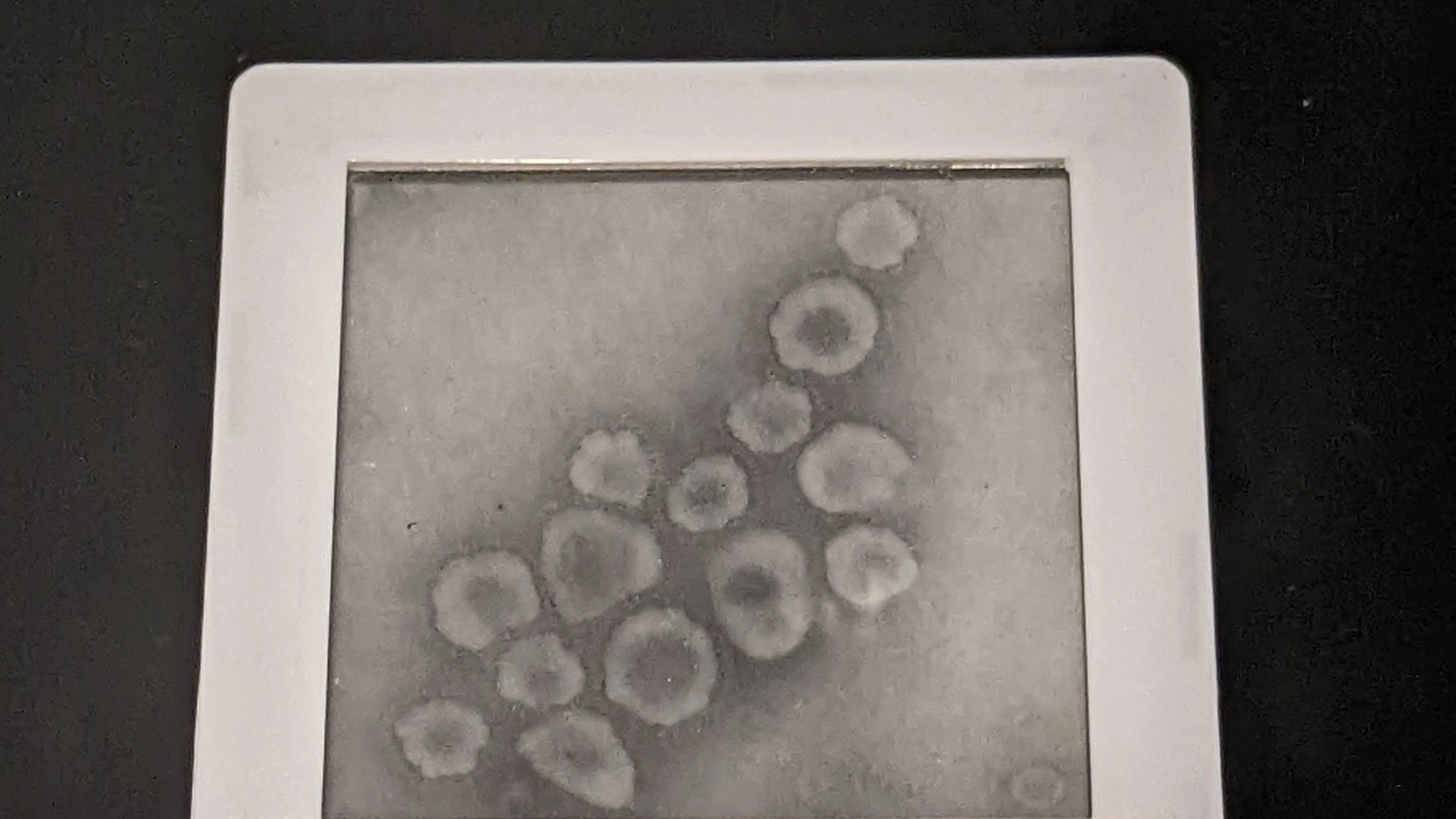
Images of human coronavirus captured in 1967 by Virologist, June Almeida
Were you surprised by how well the mRNA vaccines performed?
CEPI, my organisation, made our first investments in COVID-19 vaccines 12 days after the viral sequences were released, and of the three vaccines that we first funded, one of them was an mRNA vaccine, it was a Moderna vaccine.
Before CEPI, I used to work in an organisation in the US government called BARDA [Biomedical Advanced Research and Development Authority]. The year before I came to CEPI, we were working on Zika virus. It was 2016 when the Zika virus was spreading through the Western Hemisphere, and one of the vaccines that we had funded in 2016 was a Moderna/Zika vaccine and we still don't have a Zika vaccine, and it's 2022. So the success of the mRNA vaccines against COVID-19 was by no means a sure thing.
The rapid availability of the viral sequences was also remarkable. It was like a starting pistol for the whole pandemic response, wasn't it?
It was a remarkable moment. It let us get off to a flying start once we had the sequences, which I think it was on January 10th or 11th, 2020. The reason that we knew that the Spike protein was the right target and the reason that we knew how to manipulate the Spike protein in a way that made the initial vaccines so effective was actually because of a lot of prior investment. There had been a lot of work done to figure out how to develop vaccines against coronaviruses, because we had had prior experience with SARS-CoV-1, which had a mortality rate of 10%, and MERS-CoV, which had a mortality rate of around 35%.

Genetic sequence of the COVID-19 Spike protein
Thank goodness COVID-19 didn't have the MERS mortality rate, that would have knocked over civilization, wouldn't it?
MERS-CoV is effectively 100 times as lethal as COVID-19. And I think as we go forward and think about being prepared for future threats, we have to remember that there are hundreds of coronaviruses that have been documented in animal populations. And some of them have the lethality that SARS-CoV-1 and MERS-CoV have and others are as contagious as COVID-19 and the other cold viruses.
So the potential for a truly civilization shattering event is not a fiction. We don't know what's out there. We don't know what it would take. But we understood the potential threat posed by coronaviruses as a viral family. We knew that before COVID-19 emerged and so we hit invested in figuring out how to develop vaccines against COVID-19 on technology platforms like mRNA, but also the ChadOx1 platform that Oxford was working on.
In fact, CEPI was funding the Oxford group to develop a MERS vaccine as early as 2018. So there was a known set of possible approaches that lend themselves to a rapid response and we had done the work to understand the virus and we had shown how to develop vaccines against similar viruses on the platforms. If the world was fated to have a pandemic with a virus other than flu, we were very, very fortunate that it was a coronavirus. And we were very fortunate that it only had the lethality that COVID-19 has, as opposed to having the lethality of SARS-CoV-1 or MERS.
How fair do you feel that the distribution of COVID-19 vaccines ended up being at the end of the day?
Was it fair? No, it wasn't fair. Was it predictable? Absolutely. Early on, CEPI recognised the likely distributional challenges and focussed a lot of energy on doing everything that we could offset them. I think we were partially successful, and, and partially not.
The reason it was predictable was because we had seen exactly the same outcomes a decade or so ago when, during the last pandemic of H1N1 Swine Flu in 2009, there were 10 countries that had seriously planned for pandemics and had effectively put in place contingent purchase contracts and bought up the entire global supply of vaccine.
So there was no vaccine for the other 184 countries. In 2009 and 2010, there was an effort to share vaccines, and it got started late. The rich countries basically agreed to donate 10% of their supply of vaccine to the rest of the world. They made that announcement in September, 2009. By the time they cancelled the programme a year after they made the announcement, they had only shared 75 million doses with the rest of the world.
History showed that if we just let the market play itself out and let the countries with resources do what they would naturally do to protect their own populations, it results in no vaccine for the rest of the world.
So, as early as January, 2020, CEPI and Gavi, the vaccine Alliance started planning for how we could secure a vaccine for the rest of the world. We set up a mechanism called COVAX. And, over time COVAX has had a huge impact. We have delivered over 1.8 billion doses of vaccine 246 countries. It accounts for about 80 to 90% of the vaccine that has been administered in low-income countries. But the but the rates of delivery were much slower, because by the time we set COVAX up, by the time we secured funding to support vaccine procurement, the rich countries had bought up all of the supply.
One of the main drivers of the inequities that we saw during the rollout of COVID-19 vaccines in 2021, is the fact that vaccines are really only manufactured in a few of locations around the world. They're manufactured in the US, in the UK, in Europe, in China, and in India. And those are all regions that have very large populations. For national political leaders it is not feasible that they will give away vaccine when their own populations haven't been served. So there were structural causes of the inequity, in addition to lack of financing, problems with distribution, inadequate vaccine-delivery systems in poor countries.
Ultimately, as long as there's scarcity, you'll have inequity. To address the issue of scarcity of vaccine supply, we're calling for an expansion of manufacturing capacity to regions that currently don't have much vaccine manufacturing, like Africa. If you have more centres of vaccine production, and they can scale-up quickly, you can get over the period of scarcity quickly. But those vaccine makers have to have access to the technologies like mRNA, but not limited to mRNA, which can be scaled up rapidly. An important part of being globally prepared is to prepare vaccine makers globally to have the ability to use these new technologies to rapidly produce vaccines once we identify candidate vaccines. So once we have the vaccine recipe, we can distribute it to manufactures around the world. This will speed up vaccine delivery to regions with historically limited manufacturing capacity so that they can focus on the most highly vulnerable people. And it will allow us to save lives and reduce impact of a pandemic.
In CEPI's "What Will it Take" report, it outlines five key steps that the world will have to take if we're going to be to deliver vaccines at pandemic speed. Talk us through those steps.
We need better techniques for early detection of viral threats. We need stronger norms around vaccine sharing. We have to do as much of the work in advance before the next virus emerges. We did that for SARS-CoV-1 and MERS-CoV before COVID-19 emerged. We have to do that comprehensively for as many of the viral families as we can.
We've had two pandemics now in 13 years. And one of them happened to be very, very mild, and one of them was COVID-19. Can you imagine having two COVID-scale events in a decade? We need to be prepared for the range of possible threats, we need to do the work in advance, we need to take the kinds of innovations that were developed in 2020 and optimise every step of the development. And we need to have prepared regulatory systems that are ready, that have as much information in advance as possible about the vaccine platforms, about the different viral families, about the exemplar vaccines, so that they are ready to authorise vaccines on a risk-adjusted basis.
Your report also talks about the need to speed up identification of immune response markers. Talk us through why understanding this is so important.
If you can learn how a vaccine works and look for markers in the blood that are linked to the protection that the vaccine provides, you can identify a correlate of protection. If you can produce that correlative protection with a new vaccine against the related virus, you have a pretty strong basis for believing that the vaccine will work without having to do full phase 3 testing.
We already do this with flu vaccines. Every year we develop new flu vaccines using the current technology. We don't do full phase 3 testing. And the reason we don't is because the egg-based production system for flu vaccines is very well understood. The general safety of these egg-based vaccines is well understood, we know what the correlates of protection are a certain marker of antibodies against the hemagglutinin protein which is found on the flu virus.
If regulators are comfortable with the level of hemagglutinin antibodies that are produced by a flu vaccine candidate, that they are at the level needed to provided protection, then they appprove the vaccine and release it. The vaccine testing and approval process for flu vaccines is very compressed and we need to be able to do that for many, many more kinds of viruses.
If we have been able to develop the COVID-19 vaccine within 100 days and deliver it by mid-April, 2020, how would the pandemic have played out differently?
If we'd had a vaccine in mid-April, we would have been able to deliver doses when there were about 2.2 million cases of disease rather than in December when they were close to 70 million. Of course, vaccines aren't the only solution to pandemic preparedness, and we don't claim that they are, but they're a really, really important tool. If you imagine a world that had better surveillance systems. and stronger norms around information sharing, we probably could have picked up the fact that we had a new coronavirus maybe a month earlier. And if you back the clock up another month, and we had the sequences by mid-December, 2019, rather than mid-January, 2020, then you're in a situation where you have a cumulative total of around 300,000 cases. And if you imagine a world that takes pandemic threats seriously and implements the kind of interventions locally, and for a limited frame of time, that China did by mid-January, but implemented much earlier, you're probably reducing the number of cases by another order of magnitude, and you're also reducing the geographic spread. So then you're talking about having vaccines available when there may be 30,000 cases. And all of a sudden, you're within reach of being able to prevent pandemics all together. The IMF's last estimate was that the pandemic would produce about $15 trillion of direct economic impact over several years. And that's leaving aside the economic impact of supervening crises.
Glass vial that held Margaret Keenan's first Pfizer-BioNTech COVID-19 vaccine
Do you worry that the energy might be sucked out of your pandemic plans by global complacency and other geopolitical distractions.
I'm not as concerned about complacency because I think COVID-19 hit global society like a meteorite, and I think the saliency of the threat is there. But we now have a series of cascading crises, most of which can be traced back to the pandemic, in terms of economic disruption and the impending food crisis and certainly the war in Ukraine. Political leaders have limited bandwidth and they have to focus on the crisis in front of them. And we have stacked-up crises and limited resources. I don't think any politician would deny that pandemics are an important problem. I think the argument to the political leaders has to be: look at all of the crises that we're dealing with now and remember that we're in an economic crisis because of the expenditures that we undertook during the pandemic.
We cannot, as a global society, tolerate periodic pandemics. We have the tools and technology to drastically reduce the risk of future pandemics. Pandemics are security threats. And just as with other security threats, no matter how bad the economic situation, no matter how many other crises our governments are facing, they don't stop funding their militaries. And they shouldn't stop funding things to improve health security.
So to summarise the $3.5 billion dollar price tag for CEPI's 100 day mission is a bargain, compared with the disruption that could be caused by the next pandemic?
Yes. CEPI is not claiming that we can deliver vaccines against all future threats for this price tag. This is a global project. CEPI's programme is a part of that global endeavour. We're an international organisation and we are well positioned to do certain things, especially advancing vaccine R&D. The 100 Day Mission has captured the attention of a number of very significant politicians. It has been embraced in by the UK as part of its G7 presidency, by Germany's as part of its G7 presidency, and it will be a big part of Japan's forthcoming G7 presidency. It has also been embraced at the G20. It has been written into America's pandemic preparedness plan, they have articulated that the 100-day goal is America's goal. So, you know, this is not something CEPI can do by itself. But it's something that we think we can play an important part in catalysing and in supporting.
Watch the full interview here.
Read CEPI's report on how the world can deliver pandemic vaccines in 100 days here
Images: Science Museum Group; Injecting Hope: Race for the COVID-19 Vaccine