Progress and results
CEPI is catalysing the development of vaccines and biologics against epidemic and pandemic threats, working to make them accessible to everyone who needs them and driving progress on the 100 Days Mission. Learn more about our progress and results.

Over 75
CEPI-supported vaccine candidates and platform technologies
Nearly 2 billion
COVID-19 doses delivered through COVAX, co-led by CEPI
470+
R&D partners collaborating with CEPI
Our achievements
Supporting vaccine breakthroughs
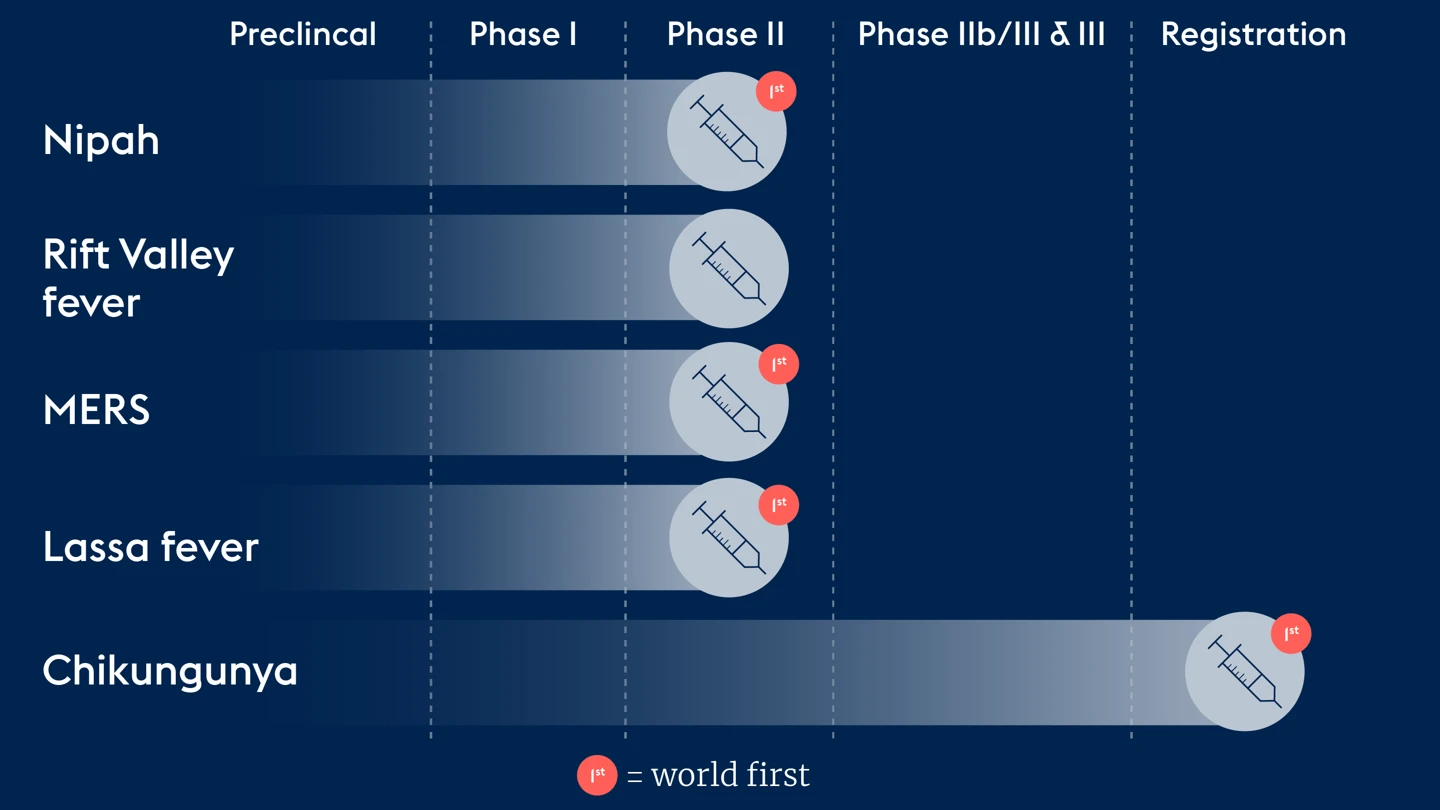
CEPI’s work has delivered world firsts for vaccines against many of its priority pathogens including the first licensed Chikungunya vaccine, the first ever Lassa fever, Nipah and MERS vaccines to reach Phase II trials and the first Rift Valley fever vaccine candidate to be studied in Phase II in an endemic region.
CEPI has also supported programmes to generate much-needed data to expand access to Ebola and Mpox vaccines.

Leading the R&D response to COVID-19
CEPI was among the first funders of COVID-19 vaccines, ultimately building one of the world’s largest portfolios, seven of which were licensed. The coalition also conceptualised and co-founded COVAX, which delivered nearly two billion doses of COVID-19 vaccines to 146 countries , saving an estimated 2.7 million lives in lower-income regions.
CEPI is the world’s leading funder of R&D into broadly protective coronavirus vaccines, investing in 13 vaccine candidates designed to counter both known and emerging threats from the coronavirus family.
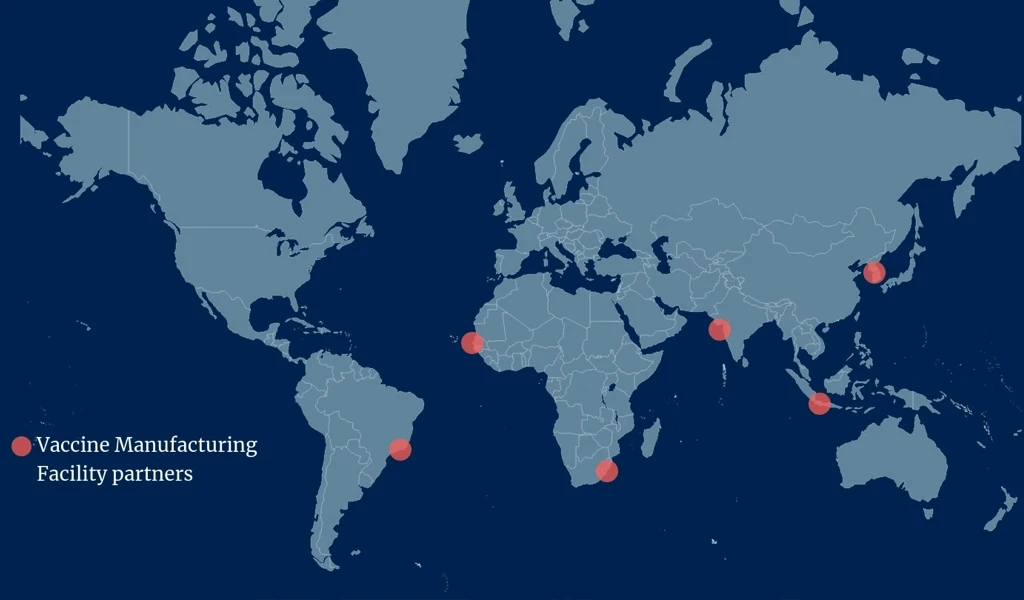
Strengthening global R&D and manufacturing networks
CEPI’s network of Global South vaccine manufacturing partners on three continents is helping to boost sustainable vaccine production and outbreak response manufacturing capacity. Its global networks of preclinical facilities and laboratories are ready to respond rapidly to outbreaks close to their source.
Driving the 100 Days Mission
CEPI has leveraged its unique role as a coalition to galvanise support and drive progress on the 100 Days Mission—a life-saving pandemic preparedness plan that is today embedded into national health security strategies around the world.
Impact in focus
Each of CEPI’s scientific projects is designed to embed speed, scale and access into new vaccine development, and behind each innovation is a team of scientists striving to better protect the world against epidemic and pandemic threats. Read our innovations for impact stories to meet them and find out about their work.
Read more
How we measure progress
CEPI’s work is guided by our updated organisational Theory of Change, which sets out how our work contributes to epidemic and pandemic preparedness and equitable response.
Our revised Monitoring, Evaluation and Learning (MEL) framework tracks progress across the full pathway from activities and outputs to outcomes and long-term impact. We measure progress through a focused set of Key Performance Indicators (KPIs) aligned to our strategic priorities, including progress toward strengthening global readiness to respond within 100 days of a new threat.
Learning is central to our approach. We regularly review performance, commission independent evaluations, and use evidence to inform strategic decisions, strengthen accountability, and improve how we work with partners.
Our Annual Progress Reports and interdependent evaluations are available at the links below.

CEPI’s achievements at a glance
CEPI has catalysed action, from product and platform development to enabling sciences and manufacturing innovations, working worldwide to accelerate the development of vaccines for those who need them most.
Download this infographic to explore CEPI’s major programmatic achievements from 2017 to now.

