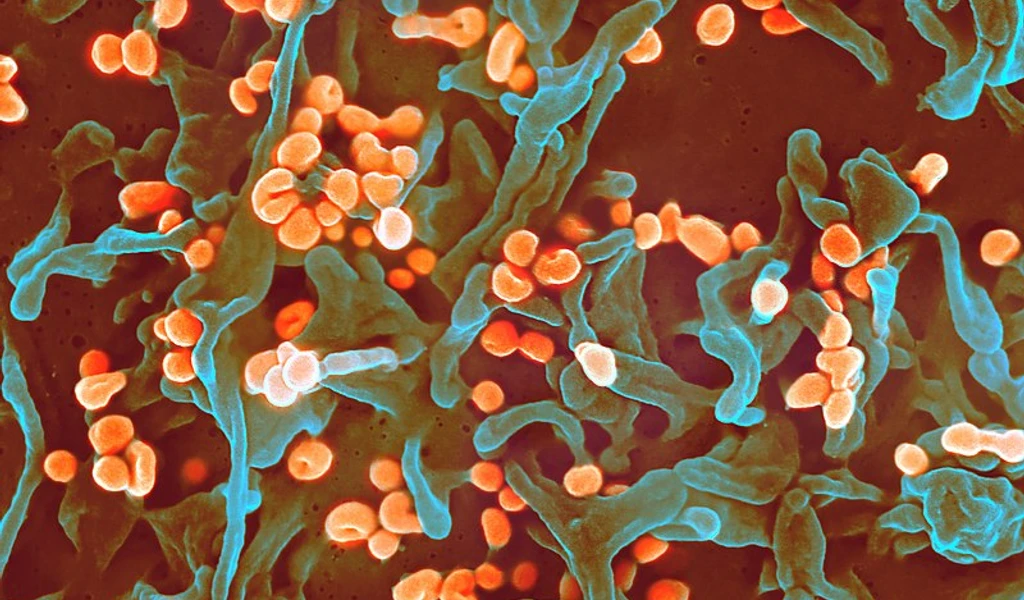
Lassa fever
CEPI's goal is to advance a Lassa fever vaccine to licensure for routine immunisation and outbreak response for affected countries. Find out more about this pathogen and our R&D efforts in this disease area.
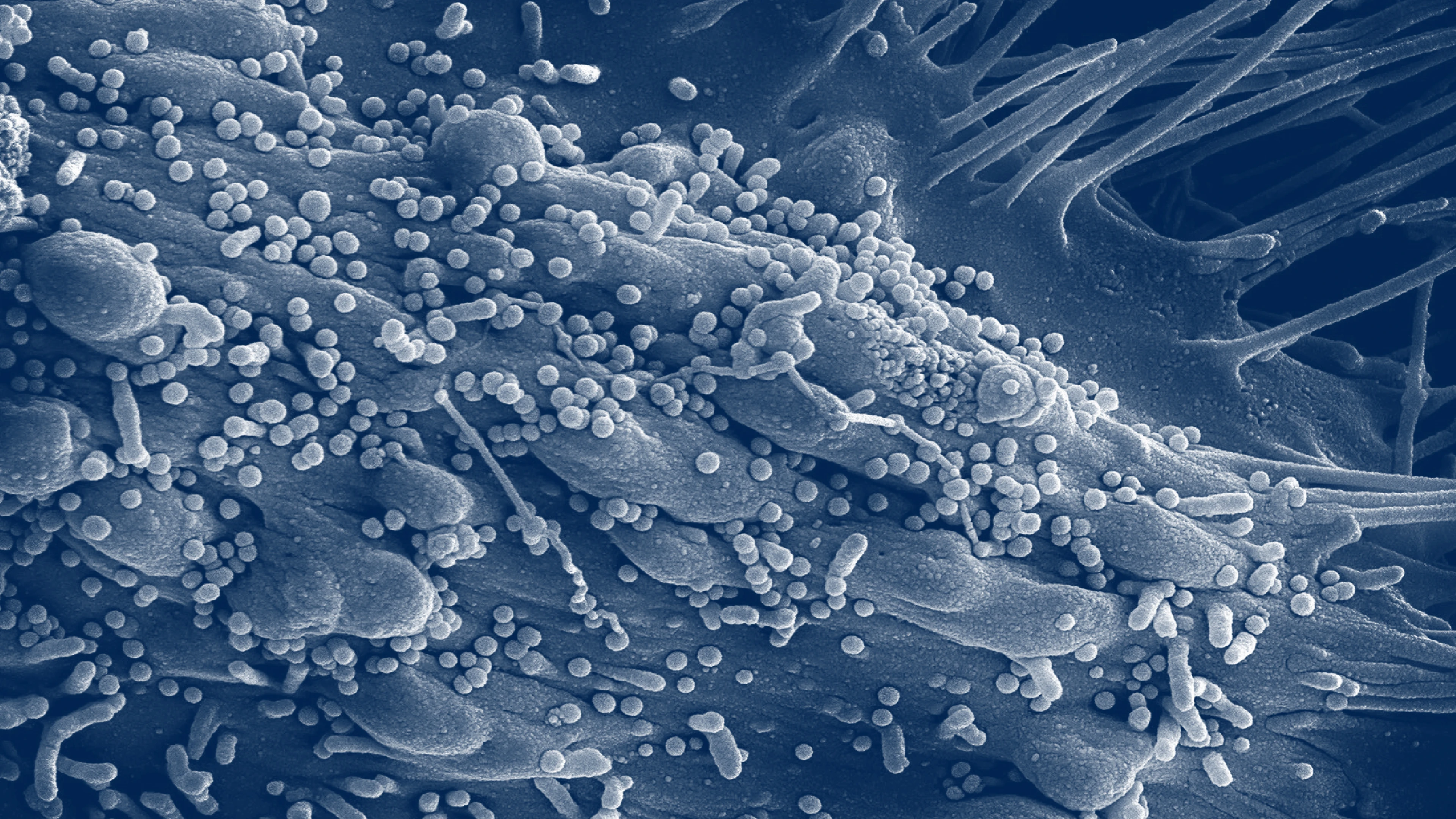
What is Lassa fever?
Lassa fever is a viral haemorrhagic disease caused by the Lassa virus. It belongs to the Arenavirus family.
Lassa virus is usually spread to people through exposure to food or household items that are contaminated with urine or faeces of infected Mastomys rats.
Person-to-person transmission may occur after exposure to virus in the blood, tissue, secretions, or excretions of an individual infected with Lassa virus.
In some regions of West Africa, where Lassa virus outbreaks are becoming more frequent, Mastomys rats are consumed as food—increasing the risk of disease transmission.
WHO has identified Lassa fever as a priority pathogen in urgent need of R&D because it poses a significant public health risk due to its epidemic potential.
No Lassa fever vaccines are currently licensed for human use.

90-95%
Number of human infections due to indirect exposure or direct contact with infected Mastomys rats
4
CEPI-funded Lassa vaccines in active development
20%
Proportion of Lassa virus infections that result in severe disease or death
Where does Lassa fever occur?
Lassa fever occurs regularly in parts of West Africa, and is known to affect people in Benin, Ghana, Guinea, Liberia, Sierra Leone, Togo, and Nigeria.
Climate change and population growth could further amplify the threat posed by Lassa fever, extending the reach the disease.
Estimates of the annual number of Lassa virus infections in West Africa range from 100,000 to 300,000.
However, these are likely to be underestimates because of challenges related to Lassa fever surveillance, such as difficulties in case detection as a result of nonspecific symptoms and limited access to specialised laboratories in affected countries.

What are the symptoms of Lassa fever?
Most people infected with Lassa virus are thought to have no symptoms.
Those who are symptomatic can suffer from mild headache and fever to more serious symptoms like vomiting, swelling of the face, pain in the chest and back, and bleeding from body parts including the eyes and nose.
It can also cause severe complications for mother and baby if pregnant women become infected, as well as leading to hearing loss in around a quarter of survivors.
On average, 1% of cases are fatal.
How is CEPI responding to Lassa fever?
Moving Lassa vaccines towards licensure
CEPI is one of the world’s leading funders of Lassa fever research.
Our goal is to support the development of one or more Lassa fever vaccines through to licensure, working with our partners in West Africa and across the world.
CEPI has invested in the development of six vaccine candidates, four of which are in active development.
One of CEPI’s partners, IAVI, has launched the first-ever Phase II clinical trial of a Lassa vaccine in Abuja, Nigeria.

The largest ever study of Lassa fever
CEPI created and funds Enable, the world’s largest ever study of Lassa fever. Over 23,000 participants have taken part across West Africa.
Data from this study will give researchers a better picture of the true disease burden and the number of people who are at risk of Lassa virus infection and who might benefit from Lassa fever vaccination.
Findings from the study will also guide the design of future vaccine trials and could help define future vaccination strategies when a Lassa fever vaccine has been approved for use.

Strengthening regional collaborations in affected countries
Regional and national leadership from Lassa-affected countries are critically important to advance the development of vaccines that protect against the disease.
CEPI maintains close engagement with institutions including the Economic Community of West African States (ECOWAS) Regional Centre for Surveillance and Disease Control, as well as national public health agencies, such as the Nigeria Centre for Disease Control (NCDC), and Ministries of Health in the most affected countries.
CEPI is also coordinating the ECOWAS RegECs Project, a collaboration between regulators, ethics committees, and the African Vaccine Regulatory Forum (AVAREF) network to support efficient management of clinical research in West Africa and to create a Lassa disease-related regulatory and ethics network.

On-the-spot Lassa diagnostics
CEPI is funding a four-year project led by FIND to examine and evaluate all available point-of-care testing options for Lassa as well as Nipah, another CEPI priority pathogen.
High quality diagnostic tests are essential both for early pathogen detection to stop an outbreak and as part of the vaccine development process particularly for the optimal design and conduct of clinical trials.
FIND researchers will identify the criteria for an optimum rapid Lassa diagnostic test and select rapid tests to assess those criteria. Successful diagnostics will be progressed to licensure for widespread use.

Lassa fever news

Prepare to be prepared: leveraging West Africa's research ecosystem to strengthen outbreak response

As CEPI ramps up vaccine research, a new study warns Lassa fever is set to expand its reach across Africa

Largest-ever Lassa fever research programme launches in West Africa

Themis Bioscience and CEPI Announce Initiation of Phase 1 Clinical Trial with Lassa Fever Vaccine
CEPI partner Inovio Advances Lassa Fever Candidate Vaccine Into a Clinical Trial